Medicine recalls
|
| |
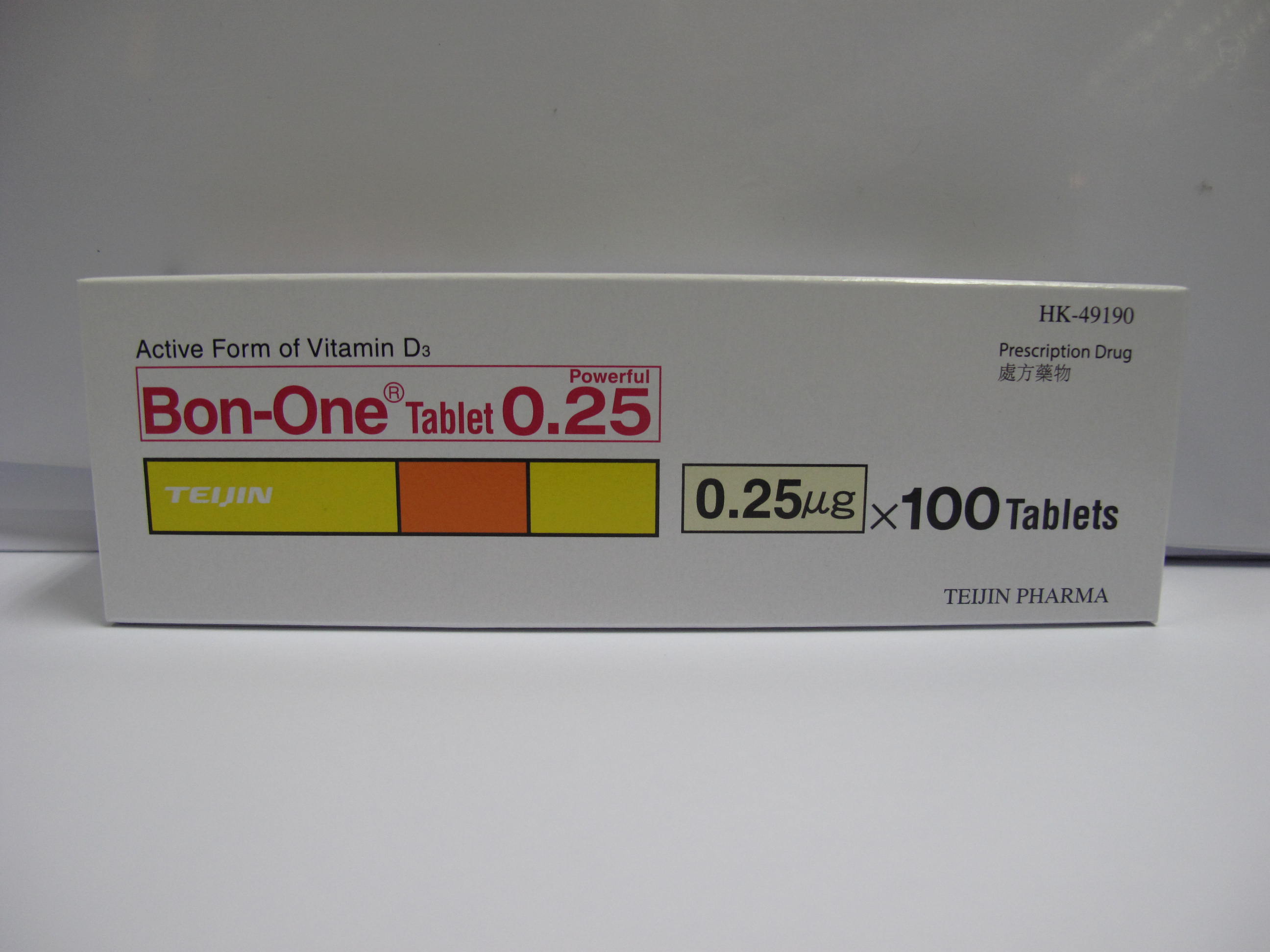
| DH endorses a batch recall of Bon-One Tab 0.25mcg |
| |
The Department of Health (DH) today (July 20) endorsed a licensed drug wholesaler, Hind Wing Co. Ltd., to recall one batch (batch number: 5125) of Bon-One Tab 0.25mcg (registration number: HK-49190) as a precautionary measure.
The DH was notified by Hind Wing that the manufacturer of the product in Japan found that one 0.5mcg tablet engraved with “0.5” was packed in a blister pack of the concerned batch of the Bon-One Tab 0.25mcg product.
Bon-One Tab 0.25mcg, containing alfacalcidol, is a prescription-only medicines used for treatment of symptoms associated with abnormal vitamin D metabolism and osteoporosis。
According to Hind Wing, about 200 boxes (containing 100 tablets per box) of the affected batch have been supplied to private doctors and pharmacies since Feb 2018.
So far, the DH has not received any adverse reaction reports related to the product. The DH's investigation is continuing.
Hind Wing has set up a hotline (2566 0562) to answer public enquiries. The DH will closely monitor the recall.
Members of the public should consult their healthcare providers if they are in doubt after taking the product.
|
| |
 | | DH endorses a batch recall of Bon-One Tab 0.25mcg | | |
|



