| |
Oral Antituberculosis Drugs
|
 |
Tuberculosis
|
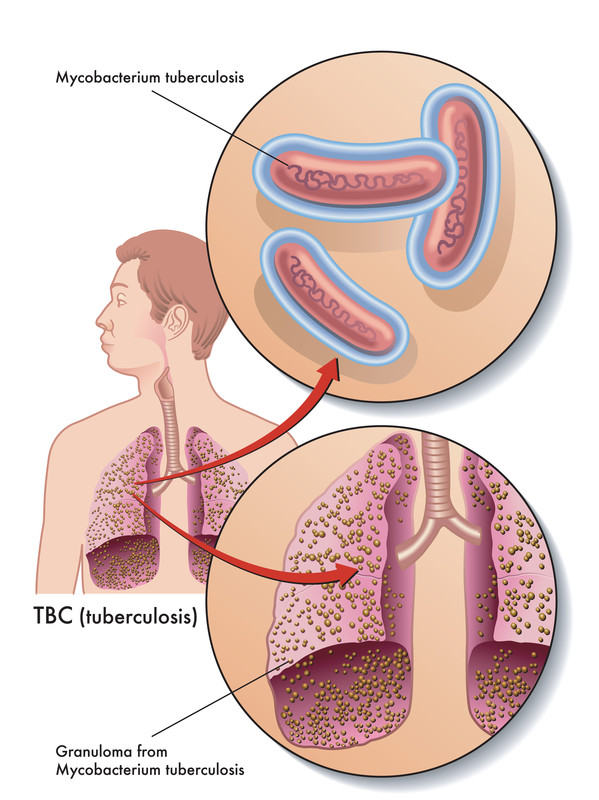
Tuberculosis (TB) is mainly caused by a bacterium called Mycobacterium tuberculosis, and occasionally by M. bovis or M. africanum. The bacteria usually attack the lungs, but TB bacteria can attack any part of the body such as the kidney, spine, and brain. If not treated properly, TB disease can be fatal.
|
TB is spread through air. When a person with TB disease of the lungs or throat coughs, sneezes, speaks or sings, they scattered the TB germs into the air. A nearby person needs to inhale only a few of these germs to become infected. The bacteria (germs) can settle in the lungs and begin to grow. From there, they can move through the blood, lymphatic system, airway or by direct extension to other parts of the body. |
In most healthy people the immune system (the body's natural defence against infection and illness) kills the bacteria and you have no symptoms. Sometimes the immune system cannot kill the bacteria, but manages to prevent it spreading in the body. This means you will not have any symptoms, but the bacteria will remain in your body. This is known as latent TB. People with latent TB infection have no symptoms, don’t feel sick, can’t spread TB bacteria to others, usually have a positive skin test reaction or positive TB blood test, may develop TB disease if they do not receive treatment for latent TB infection. |
If the immune system fails to kill or contain the infection, it can spread within the lungs or other parts of the body and symptoms will develop within a few weeks or months. This is known as active TB. Symptoms of TB depend on which part of the body is affected. Signs and symptoms of active TB include: |
- coughing that lasts more than 2 weeks,
- coughing up blood,
- chest pain, or pain with breathing or coughing,
- unintentional weight loss,
- fatigue,
- fever,
- night sweats,
- chills, and
- loss of appetite.
|
|
In general, persons at higher risk of developing active TB disease after infection include the followings - |
|
(1) |
persons who have been recently infected with TB bacteria; |
| (2) |
persons with reduced body immunity including babies and young children; |
| (3) |
persons with weakening of body immunity due to medical conditions, like HIV infection (the virus that causes acquired immunodeficiency syndrome, AIDS), diabetes mellitus, and malignancies; |
| (4) |
persons with weakening of body immunity because of taking certain medicines, like corticosteroids or other immunosuppressants. |
|
In 2014, there are 4784 of TB cases in Hong Kong. The TB cases decreases in the past 10 years from 6160 cases (year 2005) to 4784 cases (year 2014). The Tuberculosis and Chest Service of Department of Health (TB & Chest Service) provides treatment to patients free of charge. (For more information, please visit the website at http://www.info.gov.hk/tb_chest/) |
|
|
▲ back to top
Oral treatment used |
The aims of treatment of tuberculosis are to cure the disease, minimize the risk of disability or death, prevent relapse, reduce transmission of TB to others and prevent the development of drug resistance.
Treatment includes combination of drugs, which is necessary to prevent the emergence of drug-resistant bacteria. Usually, course of drug treatment lasts for 6 months.
- Initial intensive phase (four drugs for 2 months):
Four drugs (isoniazid, rifampicin, pyrazinamide, and ethambutol) are used in the first 2 months. The aim is to reduce the bacterial population as rapidly as possible and to prevent treatment failure.
- Continuation phase (two drugs for 4 months):
Two drugs (isoniazid and rifampicin) are used in the subsequent 4 months. It is to ensure that the patient is cured and does not relapse after completion of treatment.
|
|
 |
The drugs are taken either daily or three times per week as per doctor’s instruction. They have to be given under direct observation (DOT, directly observed treatment) by healthcare staff. DOT is the process, during which the TB patient takes every dose of the medication under the direct observation of healthcare staff. The healthcare staff would monitor the treatment and answer patients’ questions to enhance their compliance.
In addition to the above choice of drug, the doctor may select alternative oral TB drugs including rifabutin, para-aminosalicylic acid (e.g. sodium aminosalicylic acid (also known as sodium aminosalicylate)), cycloserine, prothionamide and levofloxacin. Some antituberculosis drugs are given by injection, such as streptomycin and kanamycin. In the case of drug-resistant TB, the duration of treatment may last for 12 to 30 months.
As inflammatory response to tuberculosis can cause considerable tissue damage and adjunctive corticosteroid therapy may be used to counter this. For more information about oral corticosteroids, please refer to http://www.drugoffice.gov.hk/eps/do/en/consumer/news_informations/dm_02.html
As a general rule, the doctor may modify the drug combinations, duration of treatment and dosages to suit the particular needs of individual patients depending on the form of TB (e.g. latent or active, relapsed, treatment interrupted, treatment failure), your life stage (e.g. children, pregnancy, breastfeeding), overall health (e.g. co-HIV infection, renal or liver diseases), possible drug resistance, medical test results (e.g. drug susceptibility test) and the site of disease in the body.
All the above mentioned oral antituberculosis drugs are registered in Hong Kong and are available in dosage forms such as capsules or tablets. They are prescription-only medicines and should be administered strictly under doctor’s instructions and recommendations.
|
|
|
▲ back to top
Common side effects and precautions
Oral Antituberculosis
Drugs
|
Common side effects
|
Precautions
|
| 1. |
Isoniazid |
- Peripheral neurotoxicity (presents as numbness or a tingling or burning sensation of the hands or feet)
- Liver damage
- Nausea
- Vomiting
|
- Should not be used in patients with drug-induced liver disease.
- Use with caution in patients with hepatic impairment. Should signs of liver disorder such as persistent nausea, vomiting, malaise or jaundice (yellowing of the skin and whites of the eyes) occur, discontinue treatment and seek medication attention immediately.
- Patient with renal impairment is at risk of ototoxicity (damage to ears) and peripheral neuropathy (damage of peripheral nerves, often causes weakness, numbness and pain, usually in your hands and feet), prophylactic pyridoxine is recommended.
- Use with caution in patients who have slow acetylator status (this means the body may process isoniazid more slowly, leading to an increased risk of side-effects). Dose adjustment may be required.
- Use with caution in patients with epilepsy, history of psychosis, alcohol dependence, malnutrition, diabetes mellitus, HIV infection and acute porphyria (porphyria is a rare illness which affects the metabolism).
- Peripheral neuropathy (a common side effect) is more likely to occur where there are pre-existing risk factors, such as diabetes, alcohol dependence, chronic renal failure, malnutrition, HIV infection, pregnancy and breastfeeding. Your doctor may give you pyridoxine prophylactically.
- Isoniazid can cause fits, mental health problems and optic neuritis (inflammation of the optic nerve that may cause loss of vision). If affected you should not drive or use machinery.
- Avoid alcohol and the following food as isoniazid interacts with them e.g. cheese (Swiss or Cheshire, red wine, fish (tuna, skipjack, Sardinella)).
|
| 2. |
Rifampicin |
- Liver damage
- Influenza-like, abdominal and respiratory symptoms, shock, renal failure and thrombocytopenic purpura (decrease of platelets present in the blood, resulting spontaneous bleeding and bruising of the skin in purple spots) on intermittent treatment
- Gastrointestinal symptoms including anorexia, nausea, vomiting, diarrhoea
|
- Avoid to use in patient with jaundice, rifamycin hypersensitivity and acute porphyria.
- Discontinue treatment and seek immediate medical attention if symptoms of liver damage e.g. persistent nausea, vomiting, malaise or jaundice (yellowing of the skin and whites of the eyes) develop.
- Should not be used in patient who are concurrently receiving saquinavir/ritonavir therapy due to an increased risk of severe hepatocellular toxicity.
- You may feel dizzy or faint, have problems with vision or have other side effects that could affect your ability to drive while taking this medicine. If this happens, do not drive or use any tools or machines.
- Use with caution in patients with renal impairment if dose is larger than 600 mg per day.
- Use with caution in patients with alcohol dependence, on prolonged therapy, has diabetes, liver impairment, are pregnant and breastfeeding.
- For women using combined hormonal contraceptive methods including pills, patches or vaginal rings should consider alternative contraceptive methods because rifampicin reduces the effectiveness of the hormonal contraceptives.
- Harmless orange-red discoloration of the urine, faeces, sweat, saliva, sputum, tears, and other body fluids. Soft contact lenses and clothing may become permanently stained.
|
| 3. |
Pyrazinamide |
- Liver damage
- Hyperuricaemia and may lead to gout attacks
- Nausea
- Vomiting
- Anorexia
|
- Avoid to use in acute gout attack and severe liver impairment.
- Discontinue treatment and seek immediate medical attention if symptoms of liver damage e.g. persistent nausea, vomiting, malaise or jaundice (yellowing of the skin and whites of the eyes) develop.
- Use with caution in patients with gout, liver impairment, diabetes, kidney impairment, porphyria, during pregnancy and breastfeeding.
- Take special care if you are taking oral contraceptives, since pyrazinamide may stop the pill from working. Use extra barrier contraception methods (e.g. condoms) whilst you are taking pyrazinamide and for one month afterwards.
|
| 4. |
Ethambutol |
- Optic neuritis (inflammation of the optic nerve), visual disturbances in the form of loss of acuity, red/green colour blindness, and restriction of visual fields
- Peripheral neuritis
|
- Avoid in patient with optic neuritis or poor vision.
- May cause eye or eyesight problems such as eye pain, visual disturbances, colour blindness, reduced vision or loss of vision – stop medication and seek immediate medical advice if you have any problems with your vision.
- Ethambutol occasionally causes sight problems and tingling or numbness in hands or feet. If this happens, do not drive or operate machinery.
- Use with caution in patients with kidney impairment, gout, visual defects, pregnant patients, elderly and children in whom evaluation of changes in visual acuity may be difficult.
|
| 5. |
Rifabutin |
- Gastrointestinal disturbance including nausea and diarrhoea
- Blood disorders (including neutropenia , leucopenia, anaemia, thrombocytopenia)
- Rash
- Myalgia (muscle pain or tenderness)
- Fever
|
- Avoid to use in patient with hypersensitivity to rifamycin, during pregnancy and breastfeeding.
- Discontinue treatment and seek immediate medical attention if symptoms of liver damage e.g. persistent nausea, vomiting, malaise or jaundice (yellowing of the skin and whites of the eyes) develops.
- Use with caution in patients with alcohol dependence, on prolonged therapy, liver impairment, kidney impairment, uveitis and acute porphyria.
- For women using combined hormonal contraceptive methods including pills, patches or vaginal rings should consider alternative contraceptive methods because rifabutin reduces the effectiveness of the hormonal contraceptives.
- Harmless orange-red discoloration of the urine, faeces, sweat, saliva, sputum, tears, and other body fluids. Soft contact lenses and clothing may become permanently stained.
|
| 6. |
Para-aminosalicylic acid e.g. Sodium aminosalicylate |
- Gastrointestinal effects including nausea, vomiting and diarrhoea; may be reduced if taken with food or with an antacid
- Hypersensitivity reactions including fever and rashes
|
- Avoid to use in patient with severe kidney impairment, during pregnancy and breastfeeding.
- Discontinue immediately if signs or symptoms of liver toxicity (including rash, fever and gastrointestinal disturbance).
- Stop taking medication immediately and seek medical attention at first signs of hypersensitivity developed. Symptoms include a rash, often followed by fever, and much less frequently, gastrointestinal disturbances of anorexia, nausea or diarrhoea.
- Use with caution in patients with liver impairment, mild to moderate kidney impairment, gastric ulcer, glucose-6-phosphate dehydrogenase (G6PD) deficiency.
- Sodium aminosalicylate contains sodium, used with caution in patients with heart failure.
|
| 7. |
Cycloserine |
- Mainly neurological, including headache, dizziness, vertigo, drowsiness, tremor, convulsions, confusion, psychosis, depression, anxiety
|
- Avoid to use in patient with epilepsy, depression, severe anxiety, psychotic states, alcohol dependence, porphyria and severe kidney impairment.
- Use with caution in patients with mild to moderate kidney impairment, during pregnancy and breastfeeding.
- You must not drink any alcohol if you are taking this medicine.
- Should allergic dermatitis or symptoms of central nervous system toxicity (such as convulsions, psychosis, somnolence, depression, confusion, hyperreflexia, headache, tremor, vertigo, paresis, or dysarthria) develops, drug discontinuation or dosage reduction may be necessary, seek medical attention.
|
| 8. |
Prothionamide |
- Gastrointestinal disturbances including nausea, vomiting, diarrhoea, anorexia, excessive salivation, metallic taste, stomatitis and abdominal pain
|
- Should be used with caution in depression or other psychiatric illness, diabetes mellitus.
- Should not be used in severe liver impairment and pregnancy.
|
| 9. |
Levofloxacin |
- Nausea
- Vomiting
- Diarrhoea
- Flatulence
- Constipation
- Hyperhidrosis
- Headache
- Dizziness
|
- Should not be used in patients with quinolone hypersensitivity, history of tendon disorders related to quinolone use, pregnancy and breastfeeding.
- Should be used with caution in patients with a history of epilepsy or conditions that predispose to seizures, G6PD deficiency, myasthenia gravis (risk of exacerbation), patients with risk factors for QT interval prolongation (changes in the electrical activity of the heart shown in the electrocardiogram [ECG] that can lead to an abnormal heart rhythm which can be fatal) (e.g. electrolyte disturbances, acute myocardial infarction, heart failure with reduced left ventricular ejection fraction, bradycardia, congenital long QT syndrome (a disorder of the heart's electrical activity that can cause sudden, uncontrollable, dangerous rate or rhythm of the heartbeat in response to exercise or stress), concomitant use with other drugs known to prolong the QT interval, history of symptomatic arrhythmias), history of psychiatric illness, renal impairment, children or adolescents.
- Exposure to excessive sunlight should be avoided (discontinue and seek immediate medical attention if photosensitivity occurs).
- Should discontinue and seek immediate medical attention if psychiatric, neurological or hypersensitivity reactions (including severe rash) occur.
- Tendon damage (including rupture) has been reported rarely. Tendon rupture may occur within 48 hours of starting treatment; cases have also been reported several months after stopping treatment. Patients over 60 years of age are more prone to tendon damage; the risk of tendon damage is increased by the concomitant use of corticosteroids; if tendinitis is suspected, levofloxacin should be discontinued immediately and patient should seek medical attention.
|
|
|
▲ back to top
General advice
|
-
Adhere to treatment and medication, lead a happy life, eat a balanced diet with adequate rest for better recovery.
- In addition to directly observed treatment (DOT), some ways to help you remember to take your medicines includes take your pills at the same time every day, ask a family member or a friend to remind you to take your pills, mark off each day on a calendar as you take your medicine and put your pills in a weekly pill dispenser.
- For most active TB patients, strict isolation is not necessary. Sick leave may be recommended to stay away from work, school or college in the initial treatment period.
|
 |
- After taking the medicines for about 2 or 3 weeks, you may no longer be able to spread TB bacteria to others. Regular cleaning of the environment should be done as general hygienic practice. The dust filters of air-conditioners should be cleansed as usual.
- Take basic precautions to prevent TB from spreading to your family and friends: wear a surgical mask, cover your mouth preferably with a disposable tissue when coughing, sneezing or laughing, dispose used tissues in a sealed plastic bag, open windows when possible to ensure a good supply of fresh air and using a fan to blow indoor air outside, not sleep in the same room with other people as you could cough or sneeze in your sleep, and observe other personal hygiene including keep hands clean and wash hands properly.
- Make sure people who may have spent time with you wear a mask and wash hands frequently. TB & Chest Service may need to test them for TB infection and to see if any preventive therapy is required. If they have symptoms suggestive of TB, ask them to seek prompt medical consultation at the TB & Chest Service.
- Clothing, beddings, kitchen utensils, and walls do not play significant role in the transmission of the bacteria. TB is not spread by shaking someone's hand, sharing food or drink, touching bed linens or toilet seats and kissing.
- After completion of antituberculosis treatment, the chance for relapse of TB should be low. However, you should still maintain good bodily health and lead a healthy lifestyle to prevent relapse of the disease. You should report for re-examination if symptoms suggesting tuberculosis recur.
- Lead a healthy lifestyle can help in preventing the development of TB disease. This includes adequate exercise, enough rest and sleep, balanced diet, avoidance of smoking and alcohol, breathing fresh air and maintaining a cheerful mood.
|
|
|
|
▲ back to top
Communication with your doctor
|
| |
 |
- Inform your doctor of your medical history including if you are HIV carrier, any previous treatment for tuberculosis, any allergies to known drugs, colourants or preservatives, as some diseases may warrant special precautionary measures.
- Antituberculosis drugs can interact with other medicines. Inform your doctor of the medicines you are taking, including prescription, over-the-counter medicines, herbal or vitamin supplements, so they can decide whether an antituberculosis medicine is safe for you to take or change the doses of medicines.
|
- Eating certain foods, using alcohol or tobacco with certain medicines may cause interactions to occur. Discuss with your healthcare professional the use of your medicines with food, alcohol or tobacco.
- Inform your doctor that you are pregnant or breastfeeding as some of the oral antituberculsois drugs should not be taken by women who are pregnant or breastfeeding.
- Have regular medical follow-up as advised by your doctor. If your symptoms do not improve within 2 to 3 weeks, or if they become worse, check with your doctor. Medical tests such as blood, urine, eye, hearing, and nerve tests may be needed to check for progress or unwanted effects. Other side effects not listed may also occur. If you notice any side effects, check with your doctor.
- Some antituberculosis drugs interfere with medical tests. You should tell the doctor in charge if you are taking antituberculosis drugs.
|
|
|
|
▲ back to top
Storage of oral antituberculosis drugs
Oral antituberculosis drugs should be kept in a cool, dry place in well-closed containers, protected from light and unreachable by children to prevent accidental ingestion. Unless specified on the label, medicines should not be stored in refrigerators.
|
|
▲ back to top
▲ back to top
|
|
Acknowledgement : The Drug Office would like to thank the Professional Development and Quality Assurance (PD&QA), and Tuberculosis and Chest Service (TB & Chest Service) for their valuable contribution to the preparation of this article.
|
|
|
|
|


